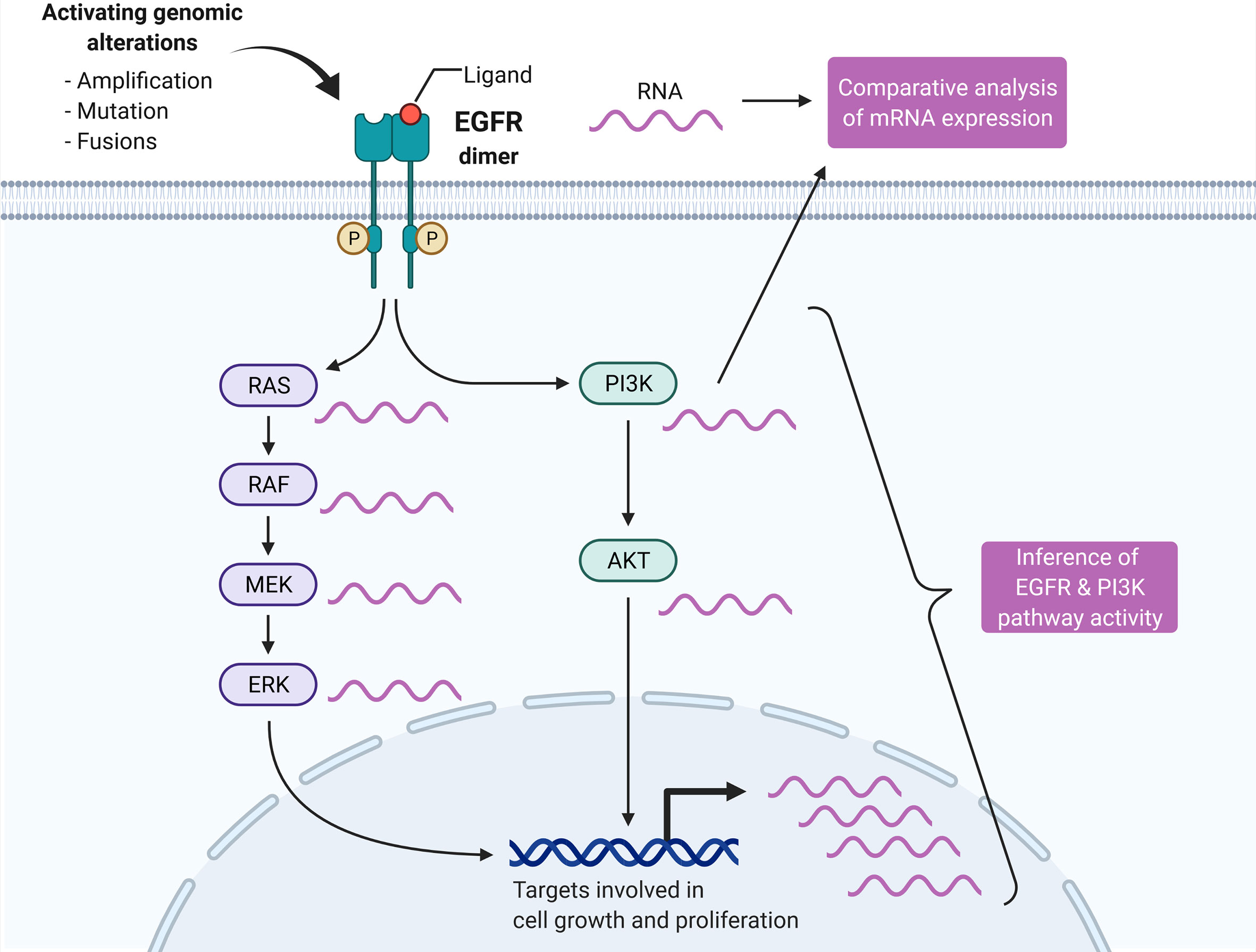  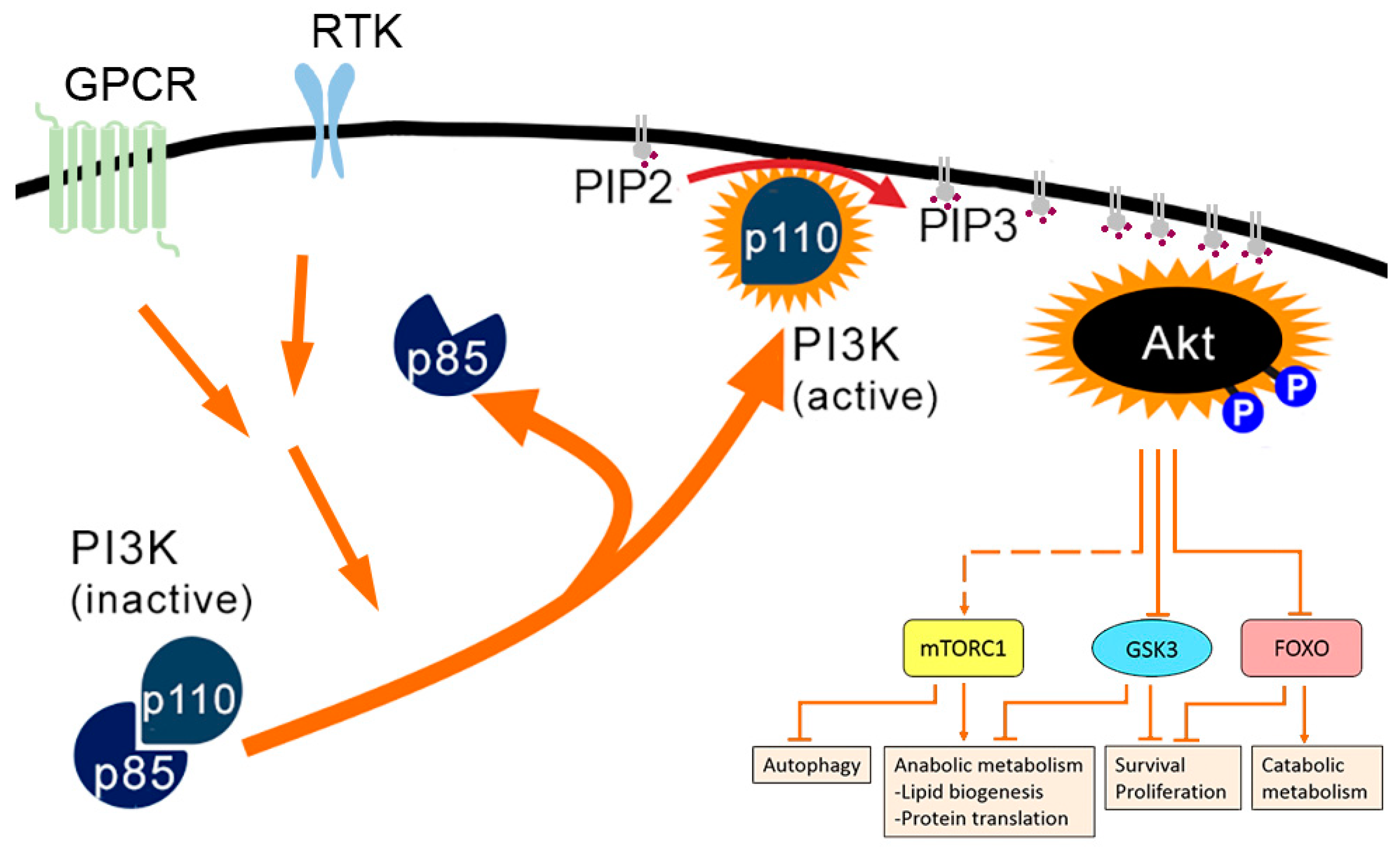
The hypothesis that each isoform recruits effector proteins to the membrane via lipid and structural reorganization ( Li and Gorfe 2012 Zhou et al. Each RAS isoform localizes to a distinct, nonoverlapping plasma membrane microdomain, where it contacts a unique pool of effector and regulatory proteins ( Parker and Mattos 2015). All these modifications are required for binding to the lipid membrane and dictate membrane localization of RAS proteins to specific microdomains. HRAS and NRAS undergo an additional palmitoylation modification ( Dudler and Gelb 1996). To become fully active, newly synthetized RAS proteins undergo a number of posttranslational modifications at the CAAX box, including the addition of a farnesyl group to the cysteine residue of the CAAX box, the cleavage of the three terminal amino residues, and, finally, the carboxymethylation of the new carboxy-terminal cysteine residue ( Tamanoi et al. RAS proteins have >80% homology sequence and differ mostly at the carboxyl terminus hypervariable region (HVR), a stretch of 25 amino acids known as the CAAX box (C, cysteine A, aliphatic amino-acid X, any amino acid) ( Parker and Mattos 2015). After receptor tyrosine kinase (RTK) engagement, RAS are activated by GEFs, which stimulate the exchange of GDP for GTP, and inactivated by GAPs that bind to RAS and stimulate its intrinsic GTPase activity. RAS proteins cycle between an inactive (guanosine diphosphate -bound) and an active (GTP-bound) state. RAS activation/deactivation cycle by guanine nucleotide exchange factors (GEFs) and guanosine triphosphate (GTP)ase-activating proteins (GAPs).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
